


Conditionnement en doses unitaires : Guide complet des systèmes et équipements à dose unique
Dans le secteur pharmaceutique et de la santé en constante évolution, la précision et la sécurité sont primordiales. Le conditionnement unitaire, également appelé conditionnement à dose unique, est devenu un pilier de la gestion moderne des médicaments. Cet article explore la définition du conditionnement unitaire, son importance, les différents types disponibles, les machines utilisées pour sa fabrication et les facteurs clés à prendre en compte lors du choix d'une solution. Nous répondrons également aux questions les plus fréquentes en fin d'article.
Le conditionnement unitaire consiste à emballer et sceller individuellement chaque dose d'un médicament. Imaginez un comprimé sous blister individuel ou un petit sachet contenant une dose précise de médicament liquide. Chaque emballage est clairement étiqueté avec les informations essentielles telles que le nom du médicament, son dosage, le numéro de lot et la date de péremption. Cette méthode évite de compter les comprimés dans un emballage en vrac, garantissant ainsi aux patients et aux professionnels de santé l'administration de la dose prescrite au moment opportun.
Initialement conçu pour les pharmacies hospitalières afin de rationaliser la distribution des médicaments et de minimiser les erreurs, le conditionnement en doses unitaires s'est désormais étendu aux établissements de soins de longue durée, aux pharmacies de détail et même aux produits de santé grand public. Son principe de base est simple : une dose, un emballage, une utilisation.
Les avantages du conditionnement unidoses ne se limitent pas à la praticité. Voici les principaux atouts qui le rendent indispensable dans le secteur de la santé :
Chaque dose étant étiquetée et scellée individuellement, le risque d'erreurs médicamenteuses — comme l'administration d'un médicament ou d'un dosage erroné — est considérablement réduit. Les infirmières et les patients peuvent ainsi vérifier rapidement le médicament avant son administration.
Les emballages unidoses protègent leur contenu des contaminants tels que la poussière, l'humidité et les microbes. Toute altération ou ouverture accidentelle est immédiatement détectable, offrant ainsi une sécurité renforcée.
Les doses prédosées éliminent les approximations et préviennent les sous-dosages et les surdosages. En cas de modification de la prescription d'un patient, les doses scellées non utilisées peuvent être retournées ou réattribuées, ce qui réduit le gaspillage de médicaments.
Les plaquettes thermoformées calendaires ou les sachets horodatés aident les patients à suivre des traitements médicamenteux complexes. Des repères visuels permettent de vérifier facilement si une dose a été oubliée, favorisant ainsi l'observance.
Légers et compacts, les emballages unidoses sont parfaits pour les personnes actives. Les voyageurs, les patients en ambulatoire et les personnes âgées apprécient leur format pratique qui se glisse facilement dans une poche ou un sac.
Les matériaux à haute barrière, tels que le papier aluminium et les plastiques spécialisés, protègent les médicaments de la lumière, de l'oxygène et de l'humidité, préservant ainsi leur efficacité tout au long de leur durée de conservation.
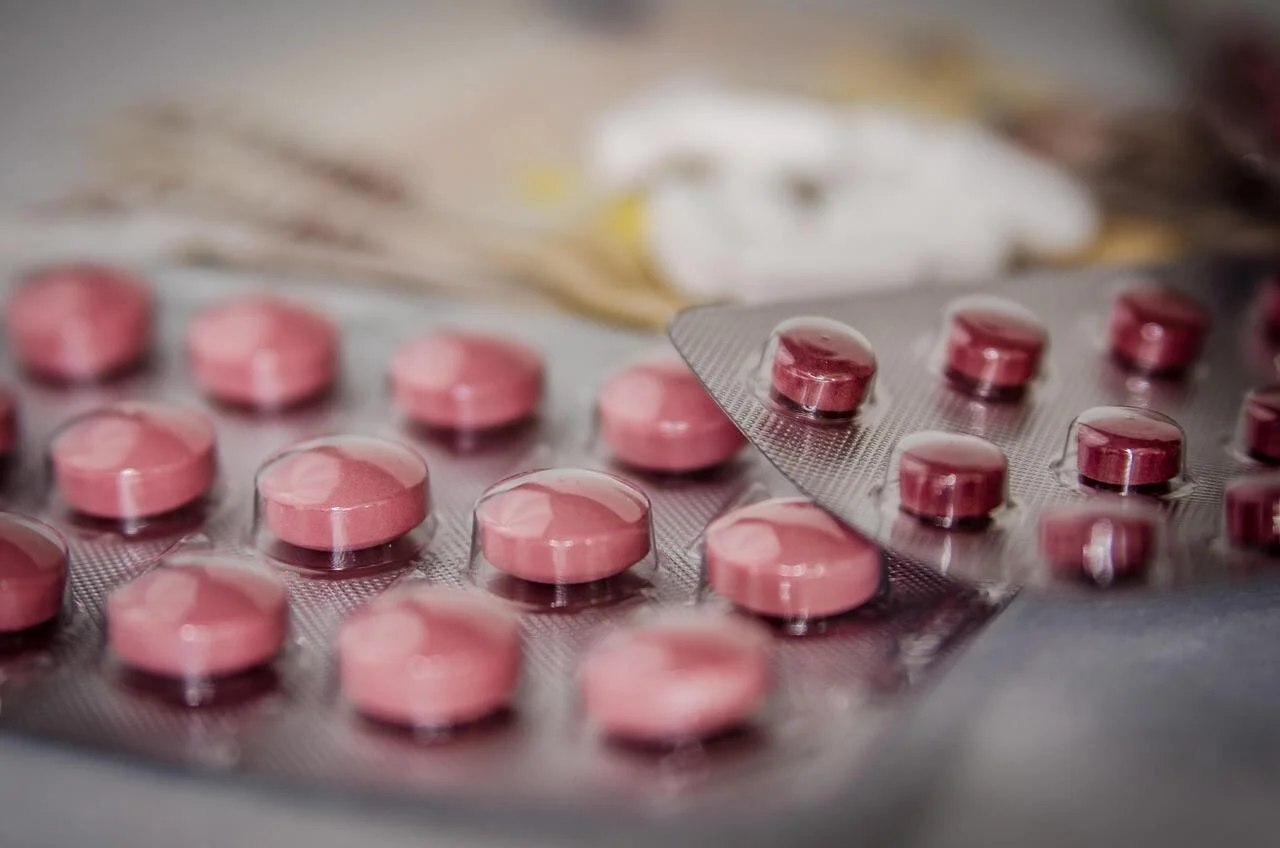
Selon la forme physique du médicament (solide, liquide, poudre ou injectable), différents conditionnements sont utilisés. Voici les plus courants :
| Type d'emballage | Description | Applications typiques |
|---|---|---|
| Plaquettes thermoformées | Cavités rigides en plastique obtenues par thermoformage, scellées par un support en aluminium ou en papier. Chaque cavité contient un comprimé ou une capsule. | Comprimés, gélules, formes orales solides. Souvent utilisés en plaquettes calendaires. |
| Bandes de conditionnement | Sachets souples obtenus par scellage de deux couches de film autour de chaque dose. Plus compacts que les blisters, ils sont souvent utilisés pour les plaquettes unitaires. | Comprimés, gélules, lorsque le gain de place est important. |
| Sachets et plaquettes | Petits sachets scellés, fabriqués à partir de rouleaux de film par des machines de formage-remplissage-scellage. Idéaux pour les poudres, les granulés ou les liquides. | Poudres orales, granulés effervescents, compléments liquides. |
| Flacons et flacons unidoses | Petits récipients en verre ou en plastique munis d'un bouchon inviolable. Utilisés pour les liquides ou les produits stériles. | Gouttes ophtalmiques, solutions injectables, solutions buvables. |
| Ampoules | Récipients en verre scellés, à ouvrir en brisant le goulot. Garantissent la stérilité des médicaments injectables à usage unique. | Vaccins, solutions injectables. |
| Seringues et stylos préremplis | Dispositifs prêts à l'emploi contenant une dose unique de médicament. Courants dans les produits biologiques et les vaccins. | Insuline, vaccins, produits biologiques. |
Chaque format garantit que la dose reste isolée et protégée jusqu'à son utilisation. Le choix dépend des caractéristiques du médicament, de la durée de conservation requise et de la voie d'administration.
La production d'emballages à dose unitaire nécessite des machines spécialisées qui automatisent les processus de formage, de remplissage et de scellage. Voici un aperçu des principaux types d'équipements :
Ces machines forment des blisters à partir d'une feuille de plastique, les remplissent de comprimés ou de gélules et les scellent avec un opercule. Deux modèles courants sont :
Machines à mouvement intermittent :Convient aux vitesses plus faibles et aux changements de format fréquents.
Machines à mouvement continu :Lignes à grande vitesse pour la production en grande série, souvent intégrées aux encartonneuses.
Similaires aux machines à blisters, elles produisent des emballages plats et souples. Elles insèrent les comprimés entre deux couches de film et scellent chaque dose. Certaines machines à blisters peuvent passer en mode plaquette.
Ces machines utilisent une technologie de formage-remplissage-scellage verticale ou horizontale pour créer des sachets à partir de rouleaux de film. Elles intègrent des systèmes de remplissage tels que des doseuses à vis pour les poudres ou des pompes à piston pour les liquides. Très polyvalentes, elles peuvent produire des sachets de différentes tailles.
Pour les liquides, les petits flacons sont remplis à l'aide de pompes doseuses de précision, puis bouchés et scellés. Le scellage par induction peut être utilisé pour garantir l'inviolabilité. Les compteurs automatiques peuvent traiter les doses solides en flacons.
Conçues pour les produits injectables stériles, ces machines remplissent les ampoules de liquide puis scellent l'extrémité à l'aide d'une flamme ou d'un laser. Elles fonctionnent dans un environnement contrôlé afin de garantir la stérilité.
Ces lignes de production complexes permettent de remplir les corps de seringues, d'insérer les bouchons et de fixer les protections d'aiguilles. Elles sont indispensables pour les médicaments biologiques et les vaccins, pour lesquels un dosage précis et une stérilité absolue sont essentiels.
Lors du choix des machines, tenez compte de la forme du produit, de la vitesse de production requise et de la compatibilité avec les processus en aval tels que le conditionnement ou l'étiquetage.
Le choix du format d'emballage et de l'équipement appropriés implique l'évaluation de plusieurs facteurs :
Forme posologique :Faites correspondre le type d'emballage au produit : comprimés/capsules → blister ou plaquette ; poudres/granulés → sachets ; liquides → flacons/ampoules ; injectables → ampoules ou seringues préremplies.
Besoins en matière de protection :Évaluer la sensibilité à l'humidité, à la lumière et à l'oxygène. Des matériaux à haute barrière comme les blisters en aluminium ou le verre ambré peuvent être nécessaires.
Volume de production :Les opérations à grand volume justifient l'investissement dans des machines à mouvement continu ; les opérations à faible volume ou les essais pilotes peuvent utiliser des équipements semi-automatiques ou manuels.
Conformité réglementaire :S'assurer que l'emballage peut porter tous les étiquetages requis (nom du médicament, dosage, lot, date de péremption) et qu'il est conforme à la réglementation pharmaceutique locale.
Coût total de possession :Il faut tenir compte non seulement du prix de la machine, mais aussi de l'outillage, du temps de changement de production, de la maintenance et du coût des matériaux.
1. Quelle est la différence entre un conditionnement unitaire et un conditionnement en vrac ?
Le conditionnement unitaire contient une seule dose par emballage, tandis que le conditionnement en vrac contient plusieurs doses (par exemple, un flacon de 100 comprimés). Le conditionnement unitaire améliore la sécurité et la précision en éliminant les erreurs de comptage.
2. Peut-on utiliser des emballages à dose unitaire pour les liquides ?
Oui. Les liquides sont généralement conditionnés en flacons, ampoules ou sachets unidoses. Par exemple, les collyres sont souvent présentés en petits flacons à usage unique.
3. Comment le conditionnement en doses unitaires améliore-t-il l'observance thérapeutique ?
En organisant les doses dans des emballages calendaires ou sur des étiquettes horaires, les patients peuvent facilement vérifier s'ils ont pris leurs médicaments. Ce repère visuel réduit les oublis de prise.
4. Quels matériaux sont utilisés dans les emballages à dose unitaire ?
Les matériaux couramment utilisés sont le PVC, le PVDC et le papier aluminium pour les blisters ; les films laminés pour les sachets ; le verre ou le plastique pour les flacons. Chaque matériau offre des propriétés de barrière différentes.
5. Existe-t-il des options durables pour le conditionnement unitaire ?
Oui, l'industrie s'oriente vers des matériaux écologiques tels que les blisters monomatériaux recyclables, les films biodégradables et une réduction de l'utilisation du plastique. Certains fabricants proposent des alternatives à base de papier.
6. Quelles machines sont nécessaires pour produire des sachets à dose unitaire ?
On utilise des machines de formage-remplissage-scellage (FFS) verticales ou horizontales. Elles forment le sachet à partir du film, le remplissent de produit et le scellent, le tout en un seul processus continu.
7. Comment puis-je garantir la conformité réglementaire des étiquettes de doses unitaires ?
L’étiquette doit comporter le nom du médicament, son dosage, les coordonnées du fabricant, le numéro de lot et la date de péremption. L’emballage doit permettre une impression ou un étiquetage clair. Consultez toujours la réglementation locale.
8. L’emballage unitaire peut-il être automatisé dans les pharmacies hospitalières ?
Absolument. Les systèmes de distribution automatisés peuvent reconditionner les médicaments en vrac en doses unitaires munies de codes-barres, et s'intégrer aux dossiers médicaux électroniques pour améliorer la sécurité.
Le conditionnement unitaire est bien plus qu'une tendance : il représente une évolution fondamentale vers une gestion des médicaments plus sûre et plus efficace. Que vous soyez fabricant de produits pharmaceutiques, pharmacien hospitalier ou ingénieur en conditionnement, comprendre les options et les technologies disponibles vous permettra de prendre des décisions éclairées, bénéfiques tant pour les patients que pour les professionnels de santé.

 Jinhai Plaza, No. 21, Jihua 5th Road, Chancheng District, Foshan City, Guangdong Province, China
Jinhai Plaza, No. 21, Jihua 5th Road, Chancheng District, Foshan City, Guangdong Province, China  +0757 82252350
+0757 82252350  +8618613054883
+8618613054883  manager@gdboanmachine.com
manager@gdboanmachine.com 

